Vexim is working on the development and commercialization of a revolutionary vertebral implant for the treatment of vertebral fractures by minimally invasive surgery. In 2017, 6 years after the commercial launch of Spinejack, Vexim achieved a turnover of almost 22 million euros. It has a 10% market share in Europe. In September 2018, the product received FDA approval for marketing in the United States. The company was sold in 2017 to Stryker for a total amount of $221 million.
See website

Synoptical view
- Truffle investment
- € 25m
In summary
Vexim is working on the development and commercialization of a revolutionary vertebral implant for the treatment of vertebral fractures by minimally invasive surgery.
As with most of the companies in its portfolio, Truffle Capital supported the company's development until the technology was commercialized. This model allows a consistent execution of the business plan, an optimal alignment of the management teams with Truffle Capital and gives the CEO the opportunity to focus his efforts on the technical and commercial success of the project.
In video
They talk about it
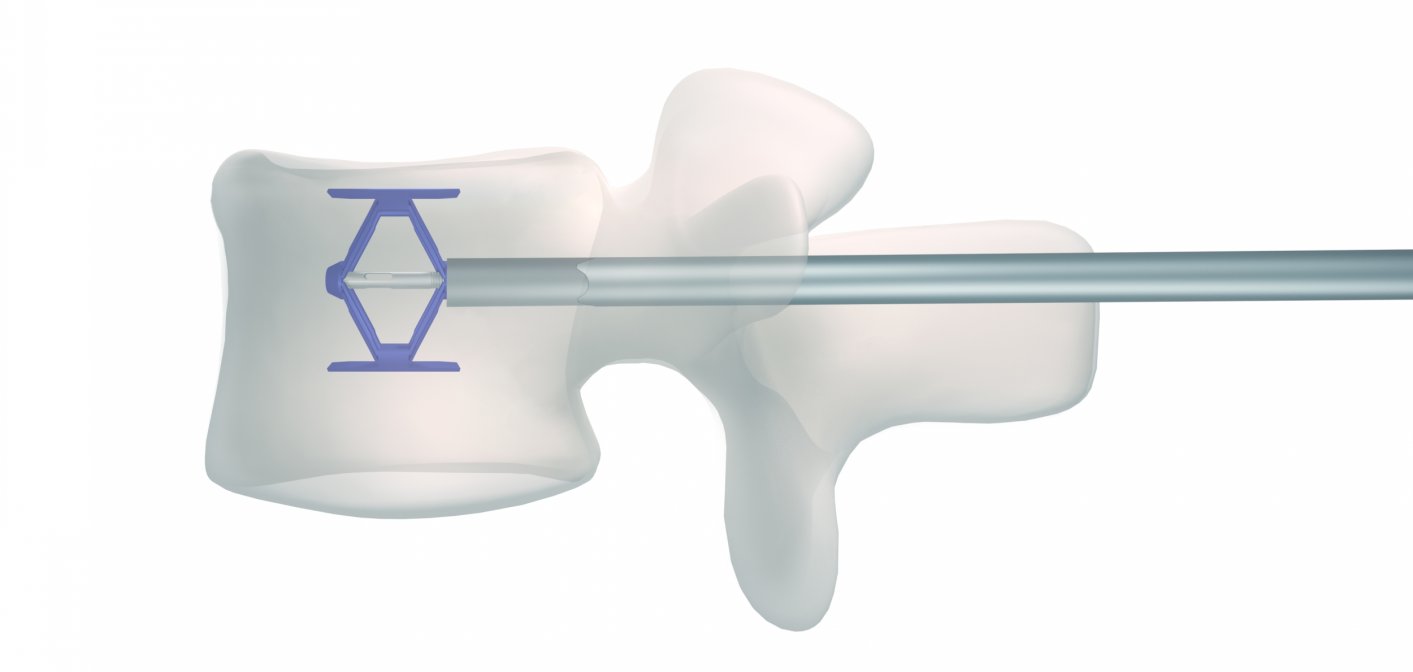
The company's flagship product is SpineJack, an implant that can repair a serious vertebral fracture in 30 minutes. A surgeon can insert this prosthesis into the spine through a simple small hole.

KEY STEPS
2009
Truffle Capital alongside Banexi finances the launch of SpineJack on the European market
2011
CE marking and marketing of SpineJack®
2012
Vexim raised €11 million for its IPO
2013
Strong adoption of SpineJack® by spine surgeons in France, Europe and other countries around the world. Vexim obtains FDA approval to market its Masterflow™ injection system in the United States
2017
Acquisition of Vexim by Stryker for $221 million
2018
Authorization to market spineJack in the United States