

ABIVAX Reports Impressive 12-month Efficacy and Safety Data from ABX464 Ulcerative Colitis Maintenance Study at United European Gastroenterology Conference
First evidence of long-term efficacy of ABX464 in ulcerative colitis
Endoscopy at Month 12 was performed in 16 / 19 patients, of whom 12 (75%) achieved clinical remission
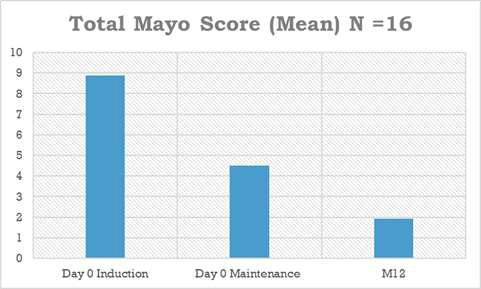
78% reduction of total mayo score, 89% reduction of endoscopic subscore and 97% reduction of fecal calprotectin biomarker (normalized)
Impressive efficacy seen in 8 weeks induction study previously reported is therefore durable or improving in this study
Continued good long-term safety profile
Ulcerative colitis Phase 2b and rheumatoid arthritis Phase 2a clinical studies ongoing, Crohn’s disease Phase 2a planned
PARIS--(BUSINESS WIRE)--Oct. 21, 2019-- Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20191020005071/en/
(Graphic: Business Wire)
ABIVAX SA (Paris:ABVX) (Euronext Paris: FR0012333284 – ABVX), a clinical-stage biotechnology company harnessing the immune system to develop novel treatments for inflammatory diseases, viral diseases and cancer, announced today new data showing that 75% of the patients were in clinical remission after a 12-month open-label oral ABX464 Phase 2a maintenance study in subjects with moderate-to-severe active ulcerative colitis (UC) who had failed immunomodulators, anti-TNFα, vedolizumab and/or corticosteroids.
The oral presentation of the data by Prof. Dr. Severine Vermeire, M.D., Ph.D., Head of the IBD Center at the University Hospitals Leuven, Belgium is taking place today at the United European Gastroenterology (UEG) Week in Barcelona, Spain during the late breaking abstracts session “IBD Highlights”.
Dr Jean-Marc Steens, M.D., Chief Medical Officer of Abivax, said: “Our new data, presented as late breaker during the “IBD highlights session“ at UEG, exceeded our expectations regarding safety and efficacy during the 12-month maintenance study. Compared to our excellent 2-month induction study data, we observed further improvements of clinical remission rates, confirmed by endoscopy, and a decrease of fecal calprotectin to normal levels. These findings confirm the potential of oral ABX464 as a well-tolerated and efficacious once-daily oral therapy for UC patients. This drug candidate has the potential to significantly improve the quality of life of patients who currently have limited treatment options in UC and other inflammatory indications, including Crohn’s disease and rheumatoid arthritis.”
The one year open-label ABX464 maintenance study was conducted in 22 patients without treatment interruption after completion of the randomised, double-blind, placebo-controlled 8 weeks induction study. A total of 19 patients completed the one year ABX464 open label maintenance study and showed good long-term safety and tolerability of 50mg given orally over 52 weeks.
At month 12, an endoscopy to assess clinical remission status (the critical parameter for regulatory authorities) was performed in 16/19 patients. During treatment with ABX464, patients reduced their total Mayo Score (mean) from 8.7 to 1.9 (-78%), their endoscopic subscore from 2.3 to 0.25 (-89%) and the fecal calprotectin biomarker (median) from 1044 microg/g to 27.9 microg/g (-97%).
Detailed analysis showed that of the 7/19 patients in clinical remission at the end of the two-month induction study, 5 patients were still in clinical remission at the end of the maintenance study and 2 patients missed endoscopy and could therefore not be assessed. Of the 12/19 patients NOT in clinical remission at the end of the induction study, 7 patients (58%) achieved clinical remission at the end of the maintenance study, while 4 patients had no remission and 1 had no endoscopy. All 3 patients without endoscopy at 12 months had fecal calprotectin levels in the normal range (<50 microg/g), which is indicative of an absence of intestinal inflammation. All 16 patients with endoscopy showed an endoscopic subscore of 0 or 1, indicative of mucosal healing and in total 12/16 (75%) of the patients undergoing endoscopy achieved clinical remission. These impressive efficacy data make ABX464 a very attractive candidate for further development. Furthermore, the data showed that ABX464 maintained the overexpression of miR124 (a critical factor of immunity and inflammation modulated by ABX464) during the 12-month study period.
Prof. Dr. Severine Vermeire, M.D., Ph.D., Head of the IBD Center at the University Hospitals Leuven, Belgium said: “The safety and durability of clinical efficacy with further improvement and increased clinical remission with longer treatment as well as significant endoscopic improvement are very promising. We are very eager to include patients in the ongoing ABX464 phase 2b study and hope to confirm the data from the phase 2a study”.
Prof. Dr. William Sandborn, M.D., Director of the Inflammatory Bowel Disease (IBD) Center at University of California (UC) San Diego Health, and Chief, Division of Gastroenterology at UC San Diego School of Medicine, added: “Only two-thirds of patients respond to currently available treatments, including biologics, and half of the responders stop responding after six to twelve months, so there is a large unmet need for effective ulcerative colitis therapies. This is a debilitating disease that greatly affects patients’ quality of life and requires expensive and cumbersome therapies. The innovative mechanism of action of ABX464 and data from this trial represent a promising new potential approach to the treatment of ulcerative colitis that could provide these patients with an easily administered, once-daily oral, long-term therapeutic management option.”
ABX464 is a highly differentiated oral drug candidate, with a novel mechanism of action based on the upregulation of a single microRNA (miRNA-124) with anti-inflammatory properties. In addition to the ongoing Phase 2b trial in UC, ABX464 is also being investigated in a Phase 2a trial in rheumatoid arthritis and soon in a phase 2a trial in Crohn’s disease, where its effects could have significant potential.
Details of the oral presentation:
Title:
Oral ABX464 QD is safe and efficacious during 52 weeks open label maintenance following a placebo controlled induction study in ulcerative colitis patients
Presenter:
Prof. Severine Vermeire, M.D., Ph.D.
Abstract number:
LB06
Location and time:
UEGweek Barcelona Fira Gran Via, Room F3 October 21 at 3:00 p.m. (CEST)
About ABX464
ABX464 was shown to exert its anti-inflammatory effects through a novel mechanism of action; it binds to the cap binding complex (CBC), which essentially sits at the 5’ end of every RNA molecule in the cell. By binding to the CBC, ABX464 reinforces the biological functions of this complex in cellular RNA biogenesis. Specifically, ABX464 enhances the selective splicing of a single long non-coding RNA to generate the anti-inflammatory microRNA, miR-124, which downregulates pro-inflammatory cytokines and chemokines like TNF-α, IL-6 and MCP-1, thereby “putting a brake” on inflammation and suggesting broad potential as a novel anti-inflammatory therapeutic agent. A seven- to ten-fold increase in miR-124 levels was observed in peripheral blood mononuclear cells (PBMCs) from healthy volunteers upon exposure to ABX464 and also in colorectal biopsies of UC patients treated with ABX464. ABX464 does not impact the splicing of cellular genes.
Webcast Presentation and Teleconference
Abivax senior management will host a webcast and teleconference Tuesday, October 22 at 2:00 p.m. CEST (Paris time) / 8:00 a.m. EST (NYC time), to discuss these clinical results and address questions. Attendees can participate by weblink (https://edge.media-server.com/mmc/p/o6m596t7) or connect by phone using the following coordinates:
Telephone conference Dial in details, Participants: Confirmation Code: 2536767 Standard International Dial-in: +44 (0) 203 0095710
Belgium
080040905
Belgium, Brussels
+32 (0) 1039 1206
China, all cities
400 608 5705
France
0805101655
France, Paris
+33 (0) 17 07 32 727
Germany
08000007416
Germany, Frankfurt
+49 (0) 6922 224 910
Japan
00531121573
Japan, Tokyo
+81 (0) 345 795 720
Netherlands
+31 (0) 2071 573 66
Switzerland
+41 (0) 445 804 873
United Kingdom
08003767425
United Kingdom
+44 (0) 8444 933 857
United States
+1 866 869 2321
United States, New York
+1 917 7200 178
About ABIVAX (www.abivax.com)
ABIVAX, a clinical stage company, is mobilizing the body’s natural immune machinery to treat patients with autoimmune diseases, viral infections, and cancer. ABIVAX is listed on Euronext compartment B (ISIN: FR0012333284 – Mnémo: ABVX). More information on the company is available at www.abivax.com/en. Follow us on Twitter @ABIVAX.
DISCLAIMER
This press release contains forward-looking statements, forecasts and estimates with respect to certain of the Company's programs. Although the Company believes that its forward-looking statements, forecasts and estimates are based on assumptions and assessments of known and unknown risks, uncertainties and other factors that have been deemed reasonable, such forward-looking statements, forecasts and estimates are subject to a number of risks and uncertainties that could cause actual results to differ materially from those anticipated in such forward-looking statements, forecasts and estimates. A description of these risks, contingencies and uncertainties can be found in the documents filed by the Company with the French Autorité des Marchés Financiers pursuant to its legal obligations including its registration document (Document de Référence). Furthermore, these forward-looking statements, forecasts and estimates are only as of the date of this press release. Readers are cautioned not to place undue reliance on these forward-looking statements. ABIVAX disclaims any obligation to update these forward-looking statements, forecasts or estimates to reflect any subsequent changes that the Company becomes aware of, except as required by law.
This press release is for information purposes only, and the information contained herein does not constitute either an offer to sell, or the solicitation of an offer to purchase or subscribe securities of the Company in any jurisdiction, in particular in France. Similarly, it does not give and should not be treated as giving investment advice. It has no connection with the investment objectives, financial situation or specific needs of any recipient. It should not be regarded by recipients as a substitute for exercise of their own judgement. All opinions expressed herein are subject to change without notice. The distribution of this document may be restricted by law in certain jurisdictions. Persons into whose possession this document comes are required to inform themselves about and to observe any such restrictions.
View source version on businesswire.com: https://www.businesswire.com/news/home/20191020005071/en/
Source: ABIVAX SA
Abivax
Communications
Pierre Courteille
pierre.courteille@abivax.com
+33 6 85 34 24 04
Public Relations France
Actifin
Ghislaine Gasparetto
ggasparetto@actifin.fr
+33 1 56 88 11 22
Investors
LifeSci Advisors
Chris Maggos
chris@lifesciadvisors.com
+41 79 367 6254
Public Relations France
Tilder
Marie-Virginie Klein
mv.klein@tilder.com
+33 1 44 14 99 96
Press Relations and Investors Europe
MC Services AG
Anne Hennecke
anne.hennecke@mc-services.eu
+49 211 529 252 22
Public Relations USA
Rooney Partners LLC
Marion Janic
mjanic@rooneyco.com
+1 212 223 4017
